All published articles of this journal are available on ScienceDirect.
Cadaveric Feasibility of Direct Intra-medullary Visualization of the Femoral Neck for Osteoporosis Research: A Pilot Study
Abstract
Introduction
Osteoporosis of the femoral neck contributes significantly to hip fracture risk and remains challenging to treat due to the limitations of systemic pharmacotherapies. We investigated whether direct intra-medullary visualization of the femoral neck is feasible and could support localized therapeutic delivery.
Methods
Four cadaveric femoral specimens were evaluated. A guidewire was placed into the femoral neck under biplanar fluoroscopy, followed by canal access using a 5 mm reamer. A 4 mm standard arthroscope was inserted, and the cavity was irrigated and debrided using a 3.5 mm shaver and a radiofrequency probe. Tetracycline and Alizarin Red dyes were instilled and visualized under ultraviolet and green light, respectively.
Results
The intramedullary cavity of the femoral neck was easily accessed and visualized. Loose cancellous bone, fibrous, and adipose tissue were observed and removed. The inferior femoral neck appeared softer than the superior portion. Bi-portal access allowed effective irrigation, visualization, and working space maintenance. Fluorochrome-based imaging revealed patchy areas of fluorescence, demonstrating potential for localized labeling and future targeted delivery. No obstruction to fluid inflow or visualization was noted.
Discussion
Intraosseous visualization enabled both structural assessment and localized labeling of the femoral neck intramedullary canal. This approach may support novel future strategies to prevent fractures of the femoral neck from osteoporosis.
Conclusion
This pilot cadaveric study demonstrates that the femoral neck intramedullary canal can be accessed and visualized using standard arthroscopic tools. This approach allows for real-time visualization and cavity preparation, providing a potential route for targeted osteoporosis therapies. Further development of endoscopic imaging systems and localized delivery agents may enable biologic reconstruction of compromised bone while minimizing systemic exposure.
1. INTRODUCTION
Osteoporosis is a critical global health concern. Its importance lies in the substantial impact on morbidity, mortality, and healthcare systems. The prevalence of osteoporosis continues to rise with an aging global population, with studies highlighting a significant increase in fracture incidence, particularly hip and vertebral fractures, which are strongly linked to morbidity, mortality, and disability [1-5].
Economically, osteoporosis imposes a profound burden, with costs estimated to escalate dramatically due to direct medical expenses, long-term care, and productivity loss [4, 6, 7]. In the U.S. alone, the cost of osteoporosis-related fractures is projected to reach $25.3 billion by 2025 [8]. Mortality following major fractures remains high, with hip fractures exhibiting up to a 20–24% one-year mortality rate [9, 10]. The number of hip fractures is projected to nearly double in the next twenty to thirty years [11].
Given its extensive health and financial ramifications, effective strategies for osteoporosis prevention, early diagnosis, and management are essential to alleviate its growing societal burden [3, 12, 13].
Current osteoporosis pharmacotherapies, while effective, are limited by class-specific shortcomings [3, 4]. Bisphosphonates (e.g., alendronate, zoledronic acid) remain first-line agents but suffer from poor patient adherence due to gastrointestinal side effects and rare yet serious complications like atypical femoral fractures and osteonecrosis of the jaw [1]. Denosumab, a RANKL inhibitor, offers potent antiresorptive effects but is associated with a rebound phenomenon characterized by rapid bone loss and increased vertebral fractures upon discontinuation [7]. Selective Estrogen Receptor Modulators (SERMs) like raloxifene provide modest vertebral fracture risk reduction but are less effective against non-vertebral fractures and increase thromboembolic risk [9]. Anabolic therapies such as teriparatide and abaloparatide stimulate bone formation but require daily injections, have duration limitations (generally two years), and have potential significant risks. The newest agent, romosozumab (sclerostin inhibitor), offers both anabolic and antiresorptive benefits but raises concerns about serious cardiovascular events [14]. In general, systemic therapies have significant limitations in terms of compliance, adherence, side effects, and time to meaningful effects.
These therapeutic gaps highlight an urgent need for next-generation options that provide sustained efficacy, biological reconstruction of bone mass, improved safety, and enhanced patient convenience.
We designed a novel approach to address this complex problem. Our hypothesis was that direct intra-medullary visualization of the femoral neck can provide a novel approach for targeted therapies of areas prone to fractures—potentially avoiding systemic side effects of pharmacotherapies while enabling anatomic cylindrical reconstruction. As a first step, a cadaveric feasibility study was conducted to prove that direct visualization was possible and to document accompanying observations.
2. METHODS
Four human cadaveric hips were examined: 86M, 82F, 77M, and one of unknown age and sex. Under plain fluoroscopy, the specimens did not appear osteoporotic. A standard arthroscope (Stryker) was used for visualization of the femoral neck intramedullary cavity. A guidewire was introduced into the femoral neck under biplanar fluoroscopy and was kept centered in both planes (Fig. 1a, b).
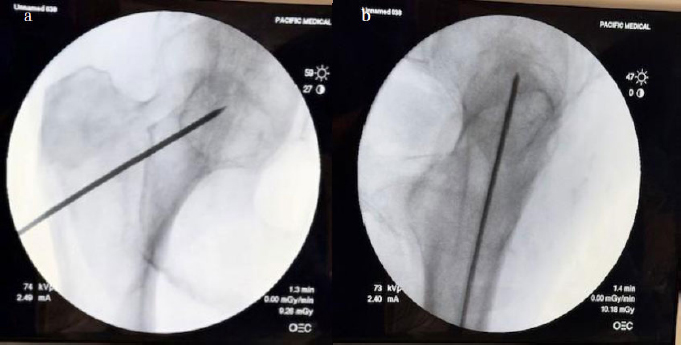
AP and lateral fluoroscopic views with guide wire. The starting point was above the lesser trochanter in the coronal plane.
Next, a 5 mm reamer was used to create a path for the 4 mm arthroscope. The cavity was irrigated, and loose cancellous bone and marrow contents were removed. A second portal was created approximately 3 mm inferior to the first (still above the lesser trochanter), through which a 3.5 mm Stryker shaver was introduced (Fig. 2).
Gentle debridement of the inferior cortex was performed. A radiofrequency probe was used to enhance debridement. Multiple images and videos were recorded, and observations were documented.
Next, 100 mg of tetracycline mixed in 30 cc of normal saline was injected into the first specimen and left for one hour. The cavity was then visualized under UV light. Subsequently, approximately 5 grams of Alizarin Red dye in 30 cc saline was instilled and examined after one hour under green light.
3. RESULTS
The femoral neck intramedullary cavity was large and easily visualized using standard arthroscopic tools (Fig. 3a, b – cavity and 3.5 mm shaver for scale). The cavity measured approximately 3 cm medial-lateral and 2.5 cm superior-inferior.
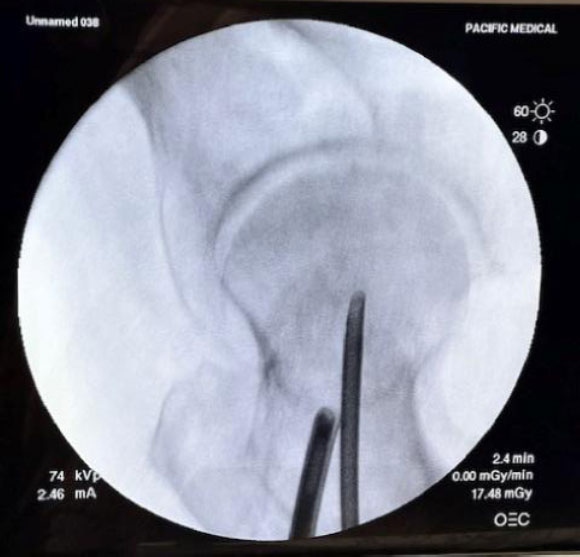
Arthroscope and shaver in place under fluoroscopy.
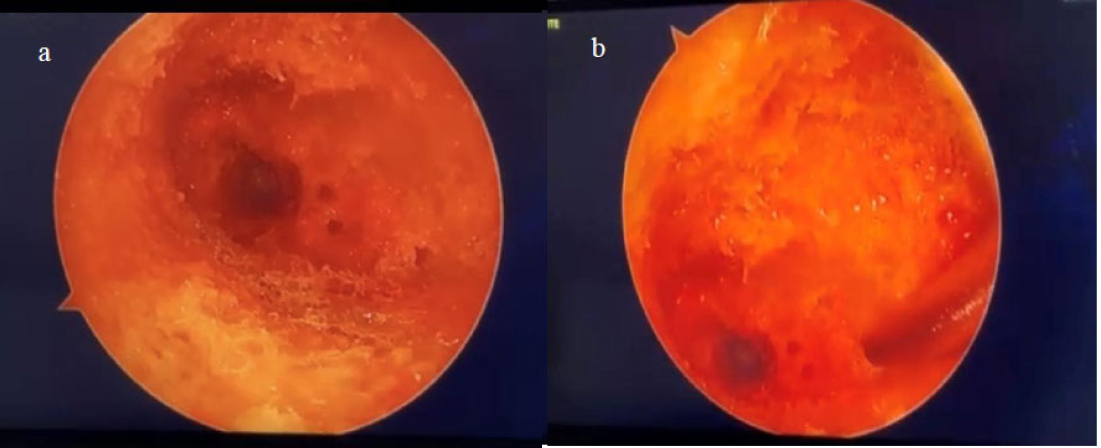
Cavity and 3.5 mm shaver for scale.
On entry, fibrofatty tissue and cancellous bone were visible (Video 1) and easily removed with irrigation and gentle debridement (Video 2). The inferior portion of the femoral neck appeared softer than the superior portion when probed. A clear transition was seen between the hollow femoral neck and the denser femoral head.
(Video 1: Upon entry into the femoral neck, the intramedullary canal, fibro-fatty tissue, and loose cancellous bone were noted)
(Video 2: Post gentle debridement, a large intramedullary canal was noted)
Fibrous bands and adipose tissue were present at the medial femoral neck/intertrochanteric junction (Fig. 4). This tissue was removed with ease using the RF probe.
After one hour, the tetracycline-infused specimen displayed patchy yellowish-green fluorescence under UV light (Fig. 5, Video 3).
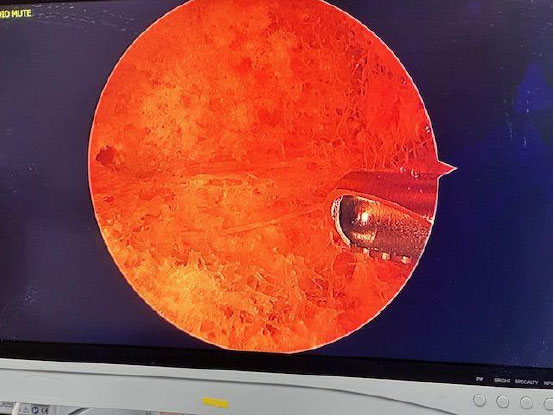
Fibrous bands at the intertrochanteric region.

Yellowish green calcification under ultraviolet (UV) light.
(Video 3: Tetracycline labelling revealed yellowish green areas of calcification under ultraviolet light)
Alizarin Red staining produced orange deposits under green light illumination (Fig. 6, Video 4). No regional dominance of labeling (superior vs. inferior) could be determined with either tetracycline or alizarin red dye.

Orange calcification deposits under green light.
(Video 4: Alizarin red staining revealed orange calcification deposits under green light in the femoral neck)
A biportal technique facilitated fluid inflow/outflow, visualization, and working space. Visualization remained unobstructed, and no fluid outflow compromise was noted once triangulation was achieved. Cortical bone thickness appeared unchanged on fluoroscopy post-debridement.
4. DISCUSSION
Bone endoscopy was first described by Harle in 1985 [15]. The technique has since been applied to osteomyelitis [15, 16], revision hip arthroplasty [17], cement removal [18], fracture reduction [19], benign tumor assessment and treatment [16, 20, 21], management of sepsis or nonunion [22], bone grafting [23], core decompression for avascular necrosis of the femoral head [24-27], canine cadaveric thoraco-lumbar studies to assess pedicle drill tracts [28], vascularity evaluation in femoral neck fractures [29], and osteonecrosis of the knee [30].
Terminology has varied, with terms like “medulloscopy” [31], “osteoscopy” [29], and “core track endoscopy” [24] used interchangeably. Both rigid and flexible endoscopes have been used successfully [31].
Previous studies have explored intraosseous approaches for osteoporosis treatment of the femoral neck using injectable materials like calcium sulfate, hydroxyapatite, growth hormone, and PMMA-based agents [32-35]. However, these approaches lack precision in replicating bone geometry and risk extravasation, resulting in non-uniform bone mass enhancement. Furthermore, no previous work has described the use of bone endoscopy for the evaluation and treatment of osteoporosis.
This pilot study demonstrated that the femoral neck intramedullary canal is easily accessible and visualizable using basic arthroscopic tools. Fibrous and fatty tissues were identified and removed. The inferior-medial neck region appeared softer than the superior, suggesting localized weakness. Spectral imaging with fluorochromes enhanced visual discrimination, aligning with prior work [36].
5. LIMITATIONS
This study was limited by a small number of cadaveric specimens and the lack of DEXA or histological confirmation of osteoporosis. Cadaveric tissue also prevented assessment of dynamic fluorochrome uptake and in-vivo biological responses. In addition, visualization challenges such as marrow turbidity and trabecular debris occasionally occurred. Although no cortical breach was seen, in-vivo use of this technique, without further refinement and biomechanical testing, could pose a risk of iatrogenic fracture. Despite these limitations, this pilot feasibility work provides a foundation for the future development of dedicated intraosseous imaging and delivery platforms.
CONCLUSION
This pilot cadaveric study demonstrates the feasibility of direct intramedullary access and endoscopic visualization of the femoral neck using standard arthroscopic tools. It enables effective tissue debridement and supports possible fluorescence-based imaging for site-specific therapeutic strategies. With further refinement, this approach could facilitate biologic reconstruction of the osteoporotic femoral neck while avoiding systemic pharmacologic exposure with its side effects.
AUTHORS' CONTRIBUTIONS
The authors confirm contribution to the paper as follows: A.Q.: Contributed to conceptualization, methodology, investigation, writing the original draft, and supervision; S.B.: was responsible for methodology, investigation, data acquisition, and review and editing; S.A.: Contributed to data interpretation, literature review, writing, and review and editing; S.H.: was involved in data curation, visualization, and manuscript formatting. All authors reviewed and approved the final manuscript.
LIST OF ABBREVIATIONS
| UV | = Ultraviolet |
| RF | = Radiofrequency |
| DEXA | = Dual-energy X-ray absorptiometry |
| SERMs | = Selective Estrogen Receptor Modulators |
| PMMA | = Polymethylmethacrylate |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Pacific Medical Inc. Bioskills Lab (Tracy, CA) provided institutional authorization for the use of donated cadaveric specimens for research and educational purposes. The work was exempt from IRB review under institutional policy.
HUMAN AND ANIMAL RIGHTS
All procedures performed in studies involving human participants were in accordance with the ethical standards of institutional and/or research committee and with the 1975 Declaration of Helsinki, as revised in 2013.
AVAILABILITY OF DATA AND MATERIALS
All data generated or analyzed during this study are included in this published article. Raw data (including additional videos and images) are available from the corresponding author on request.
ACKNOWLEDGEMENTS
We thank Pacific Medical Inc., Bio Skills Lab, Tracy, CA, for cadaveric lab support and provision of facilities used in this study.


